 2 The interpretation of CAM in this sense was stimulated by the discovery of another variant of photosynthesis, the C -pathway (see Black, 1973 Hatch and 4 Slack, 1970 Hatch, 1976). 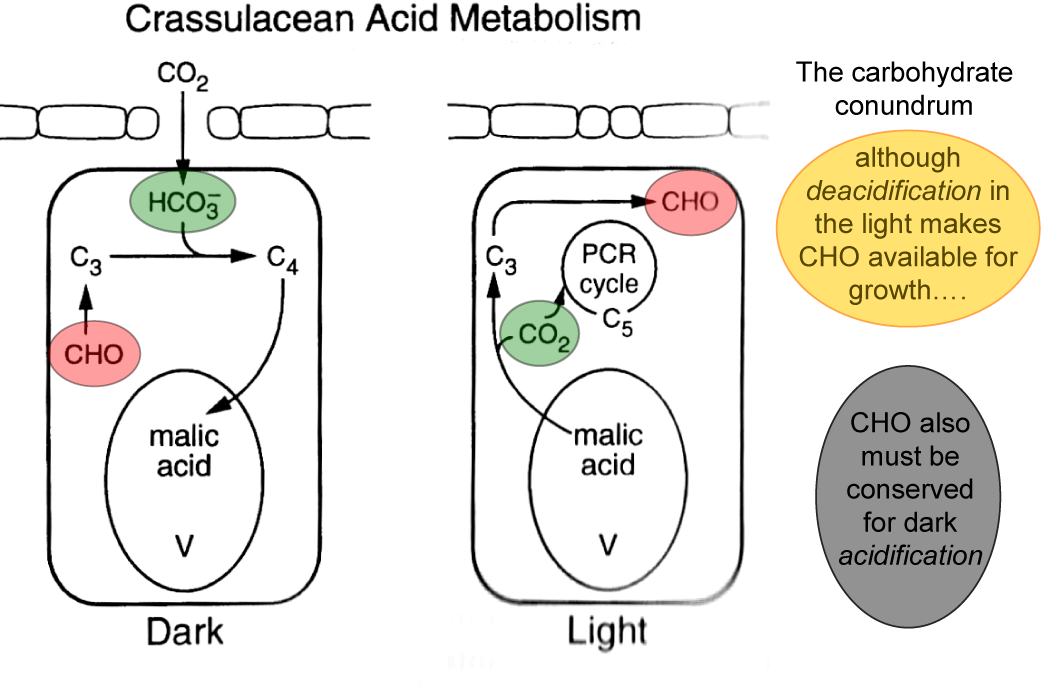
These efforts finally resulted in a clear realization that the CAM phenom enon is a variation on the mode of how plants can photosynthetically harvest CO from the atmosphere. Crassulacean acid metabolism (CAM) can be traced from Roman times through persons who noted a morning acid taste of some common house plants. Hence, it is not surprising that most research to elucidate the mechanism of CAM has been during the last 15 years since CO exchange and malate metabolism were studied and interpreted in its 2 context. Malic acid is shuttled to the vacuole of the cell (not moved out of the cell to. In CAM plants, carbon dioxide is only gathered at night, when the stomata open. PEP carboxylase still fixes CO2 to OAA, as in C4. Malic acid has two stereoisomeric forms (L- and D-enantiomers), though only the L-isomer exists naturally. It is a dicarboxylic acid that is made by all living organisms, contributes to the sour taste of fruits, and is used as a food additive. 
However, since the basic discoveries of De Saussure in 1804 that stem joints of Opuntia were able to remove CO from the 2 atmosphere during the night, and of Heyne in 1815 (see Wolf, 1960) that organic acids accumulate in the leaves of Bryophyllum calycinum during the night, the two main aspects of CAM, diurnal CO gas exchange and metabolism of malic acid, 2 have first been studied nearly independently. Cacti store the carbon dioxide they take in during the night in their cells in the form a chemical called malic acid. Malic acid is an organic compound with the molecular formula C4H6O5. Summary: The acid metabolism of certain succulent plants, now known as Crassulacean Acid Metabolism (CAM) has fascinated plant physiologists and biochemists for the last one and a half centuries. In a nutshell, 24-hour CAM occurs by phosphoenol pyruvate (PEP) carboxylase fixing CO(2)(HCO(3) (-)) over the night to form malic acid that is stored in. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
